|
3.4. The evaluation of the Vitotox test through 11 blind tested
samples
Jacky Van Gompel and Luc Thilemans
Department of Genetic and in vitro Toxicology
Janssen Research Foundation
Turnhoutseweg 30
B-2340 Beerse
Belgium.
Tel.: 0032(0)14 60 50 18
Fax: 0032(0)14 60 65 15
E-mail: This email address is being protected from spambots. You need JavaScript enabled to view it.
E-mail : mailto:This email address is being protected from spambots. You need JavaScript enabled to view it.
Summary
In order to assess the robustness, sensitivity and specificity of a recently developed screening
assay, 11 coded compounds were tested in the Vitotox assay with strains TA104recn2-4 and
TA104pr1, obtained from the Flemish Institute for Technological Research (VITO). The aim
was to assess the results from the different test systems used by the participants of this
collaborative study – Technotox - and to compare them with known literature data. The most
remarkable fact is the absence of false positive results which we also observed in our own test
panel of 45 Janssen compounds. All of the 11 samples in the current study were correctly
identified with very high sensitivity.
Introduction
New synthesis strategies together with the establishment of high throughput screening
methods in pharmacology departments are increasing the number of compounds entering the
“exploratory development” phase. Due to the necessity to optimally use resources, selections
have to be made in an early phase of development to minimise the risks of failure in later and
more expensive phases of development. Recently emerging screening tests for detecting DNA
damage and gene mutations enable the genetic toxicologist to provide crucial mutagenicity
information for selection of the most promising candidates or in priority setting between
several similar drug candidates. One of the key problems was the compound consumption and
the short time frame in which results should be obtained. Apart from the discussion about the
predictive value of bacterial tests towards human carcinogenicity, the bacterial reverse
mutation test still has a pivotal position in the final regulatory acceptance of new chemical
entities. Bacterial assays, like the SOS chromotest, based on the expression of repair genes
induced by genotoxic agents are very useful screening tools with high predictive power
towards the Ames test results. A promising new screening test, the Vitotox test developed by
VITO, which has the same basic principle as the SOS chromotest test but has the additional
advantage of the kinetic evaluation and higher dynamic range, scores very well both on
compound consumption and testing time schedule. The TA104recN2-4 strain has a lux operon
of Vibrio fisheri under transcriptional control of the recN gene, that is part of the SOS repair
system. If under the influence of a genotoxic compound the recN promotor is depressed, this
will result in expression of the lux operon and thus light production. The other strain,
TA104pr1, has the lux operon under control of a strong constitutive promotor pr1 and is used
as internal control to detect false positive compounds (compounds that act directly on the light
production (aldehydes) or enhance the metabolism). An internal evaluation study at our
laboratories with about 66 new drug candidates or drug intermediates gave very favourable
results as prediction towards Ames results (Sensitivity = 82%, Specificity = 100%,
Concordance = 92%, False negative rate = 18%, False positive rate = 0%).
Material and Methods
Chemicals
The 11 samples under investigation were distributed with a blind code. The S9-fraction from
the rat metabolic activation system was provided by Moltox. Following positive control
substances were used: 4-nitroquinoline-oxide (4-NQO) and benzo(a)pyrene (B(a)P).
Origin of the strains
Salmonella typhimurium strains TA104recN2-4 and TA104pr1 were obtained from VITO,
Mol, Belgium and were kept at –80 °C in a Biofreezer (New Brunswick Scientific ) until use.
Overnight culture
Twenty microliter of each strain (TA104recN2-4 and TA104pr1) is added to a 50 ml falcon
tube, containing 5 ml of a normal bacterial growth medium, supplemented with extra CaCl2 to
allow optimal growth. Bacterial cultures are then incubated overnight in an Innova 4000 (New
Brunswick Scientific) rotative, environmental shaker at 250 rpm and at 37ºC. A falcon tube
with only growth medium was also incubated to check the sterility of the culture medium.
One hour culture
The next morning, OD600 values were taken from the cultures. OD600 should be > 2 otherwise
incubation was continued until sufficient OD values were reached. Cultures were then diluted
10 times. 50 μl of each strain (TA104recN2-4 and TA104pr1) of the overnight culture was
added to a 50 ml falcon tube containing 2.5 ml growth medium. Bacterial cultures were then
incubated on an Innova 4000 environmental shaker at 250 rpm and at 37ºC (1 hour) to obtain
log phase growth.
Preparation of the test and control compounds
During the one hour culture period, the concentration range of the test compounds was made.
Preparation of the 96-well microtitre plate: DMSO was used as standard solvent for unknown
test compounds (1% final concentration). Environmental samples were diluted in an aqueous
buffer provided by VITO. 8 concentrations of the test compound were used together with 2
solvent controls and 2 positive controls. Serial dilutions with a factor of 2 were made. 4-NQO
was used as positive control for the test without metabolic activation in a final concentration of
4 μg/ml and benzo(a)pyrene in the presence of a metabolic activation system in a final
concentration of 800 μg/ml.
S9 mix
The S9 (batch 869) was supplied by Molecular Toxicology Inc. (NC, USA) and was stored in
a biofreezer at –80C until use. Prior to use, the S9 was combined with an Ames mutagenicity
test tablet containing the necessary co-factors such as NADP and G-6-P. The S9 fraction was
10% of the S9-mix volume. In the final measurement plate this was again 10 fold diluted to a
1% final solution. The samples without S9-mix were provided with phosphate buffer to keep
the number of bacteria the same.
Exposure
In each well of the microtitre plate, 90 μl of the one hour culture is mixed with 10 μl of the test
compound.
Luminometry
The 96-well microtitre plate was placed in an Microlumat LB96P luminometer (EG&G
Berthold) or in a Luminoskan Ascent (Labsystems) and measuring was performed with the
following parameters: 1 s/well; cycle time = 5 min; 60 cycles; incubation temperature = 30ºC.
Data handling
When the luminometer measurements were completed, data was copied and pasted into an
Excel macro sheet. The signal-to-noise ratio (S/N), being the light production of exposed cells
divided by the light production of non-exposed cells, was calculated for each measurement.
Evaluation criteria
A test compound was considered genotoxic when
· the max S/N (recN2-4)/ max S/N (pr1) > 1.5;
· A clear dose response curve is generated;
· the signal is not generated in the first 30 s;
Results
Table 1: The un-coded and grouped results for the test compound 4-nitroquinoline-N-oxide (4-
NQO).

Legend to table 1: wo_S9 : without the addition of induced rat liver S9 mix from Moltox.
w_S9: with the addition of induced rat liver S9 mix from Moltox.
Values are the induction factors as compared with the solvent controls. The red colour + bold
code represents the concentrations regarded positive.
4-NQO is a directly acting compound and this was very clearly detected in the three samples.
Only at the very high concentrations of 0.5 and 0.25 mg/ml was there some signal in the
presence of metabolic activation, probably due to overloading of the activation system. If the
sensitivity is compared between the three samples, in the first sample detection is below
0.00037 μg/ml. In sample 2 it is 0.0003 μg/ml and in sample 3 it is 0.00062 μg/ml.
Table 2: The un-coded and grouped results for the test compound 4-N-methyl-n-nitro-Nnitrosoguanidine
(MNNG).
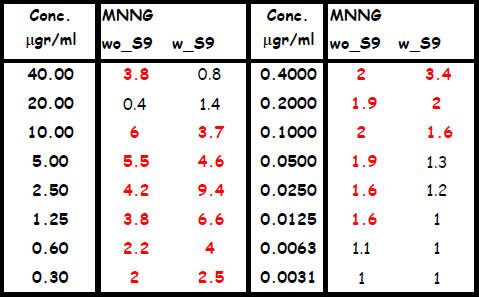
Legend to table 2: wo_S9 : without the addition of induced rat liver S9 mix from Moltox.
w_S9: with the addition of induced rat liver S9 mix from Moltox.
Values are the induction factors as compared with the solvent controls
If the sensitivity is compared between the two samples, in the first sample detection is below
0.3 μg/ml. In sample 2 it is 0.0125 μg/ml in the absence and 0.1 μg/ml in the presence of a
metabolic activation system.
Table 3: The un-coded and grouped results for test compound 2- Aminoanthracene (2-AA).

Legend to table 3: wo_S9 : without the addition of induced rat liver S9 mix from Moltox.
w_S9: with the addition of induced rat liver S9 mix from Moltox.
Values are the induction factors as compared with the solvent controls
2-AA is an indirectly acting compound which needs metabolic activation and this was very
clearly detected in the three samples. If the sensitivity is compared between the three samples,
in the first sample detection is below 0.2 μg/ml. In sample 2 it is below 0.312 μg/ml and in
sample 3 it is below 0.05 μg/ml. No activity was observed in the absence of a metabolic
activation system.
Table 4:
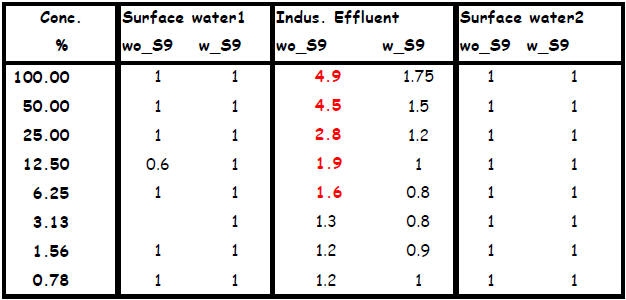
Legend to table 4: wo_S9 : without the addition of induced rat liver S9 mix from Moltox.
w_S9: with the addition of induced rat liver S9 mix from Moltox.
Values are the induction factors as compared with the solvent controls
Genotoxic activity was observed only in the industrial effluent,. In the absence of a metabolic
activation system, activity was detected from a 6.25 % dilution onwards. No DNA damaging
activity was detected in the surface water samples.
Table 5: Summary table of the lowest effect levels (LEL)
Compound code solvent Met. Act. LEL (μg/ml)
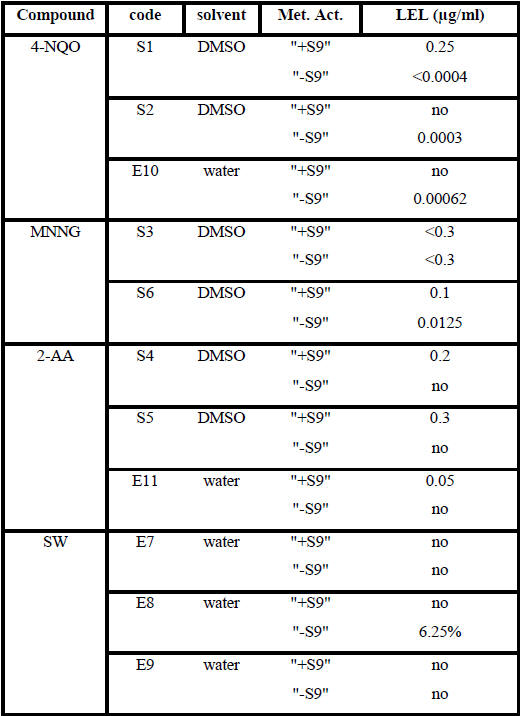
Legend to table 5: code: blind code; -S9 : without the addition of induced rat liver S9 mix
from Moltox. +S9: with the addition of induced rat liver S9 mix from Moltox. LEL values are
the calculated concentrations from the top concentrations provided. “no” means no effect
observed.
Discussion
With the currently used 96-well luminometer, two compounds can be tested in one 96-well
plate at 8 concentrations with and without a metabolic activation system using the two strains.
Hands-on time for the preparation of the test compounds is about one hour, this is more or less
the time needed for the second incubation. Results are then obtained during the 4 hours of
incubation in the temperature controlled luminometer. Data is then transferred into a specially
developed Excel spreadsheet and the final calculated data, graphs and tables are generated. For
our lab this means that one technician can handle 4 compounds a day with and without a
metabolic activation system and with all necessary controls. If we do not initiate bacterial
cultures during the weekend, we can handle 16 compounds a week. 16 additional compounds a
week can be handled per extra reading device by the same technician. By using 384-well
plates (not used for this Technotox study) and a luminometer that can handle this format, the
number of compounds can be increased to 64 compounds a week or even 80 if the incubator is
programmed for starting on Sunday and to deliver the cultures on Monday morning.
For interpretation of the results from the test compounds, all data were very straightforward.
This means very pronounced dose-response effects and a very clear discrimination between
the effects obtained in the absence and in the presence of a metabolic activation system. The
sensitivity between the three different samples can be considered as equal and no relevant
differences were found between the samples which used DMSO as solvent and the diluted
sample without DMSO. The complete range of effects was detected with our standard and
ordinary 1:2 dilution series using 8 concentrations. This means that our most diluted sample
was 128 times diluted as compared to the first concentration and 12800 times as compared to
the original solution.
The correct prediction of the surface water samples may require a different protocol as they
should perhaps be concentrated instead of diluted to detect any DNA damaging contaminants.
References
1. Gee P., D.M. Maron and B. Ames (1994) Detection and classification of mutagens: A set of base
specific Salmonella tester strains, Proc. Natl. Acad. Sci. USA, 91, 11606-11610.
2. van der Lelie D., Regniers L., Borremans B., Provoost A. and Verschaeve L. (1997) The VITO-Tox
test, a bioluminescent Salmonella typhimurium test to measure genotoxicity kinetics. Mutation Res.
389:279-290.
3. Quillardet P. and Hofnung M. (1993) The SOS chromotest: a review. Mutation Res. 297: 235-279.
4. Verschaeve L., Van Gompel J., Regniers L., Thilemans L., Vanparys P., van der Lelie D.. (1999)
The VITOTOX genotoxicity and toxicity test for the rapid screening of chemicals, Environmental
and Molecular Mutagenesis, 33, 240-248.
List of recent relevant patents
D. van der Lelie, B. Borremans, A. Provoost, L. Regniers, L. Verschaeve (1996). Novel
recombinant nucleic acid sequences, host microorganisms comprising such sequences and
use there of in tests for determining the presence of a toxic compound in a sample, for
determining both genotoxicity and mutagenicity of a sample and determining the kinetics
of genotoxicity of a sample, said method requiring luminescence measurements.
PCT/EP96/01745
D. van der Lelie, L. Regniers, S. Taghavi, P. Corbisier, L. Verschaeve (1999). Diagnostic
system and method for determining the presence of a genotoxic compound in a sample.
PCT/BE99/00049
http://www.the-scientist.com/yr2000/jan/profile_000110.html
NADP Nicotinamide adenine dinucleotide
phosphate, oxidised form
BIB3013 Assay (by titration) >98%
195 Euro / 100 mg
Glucose-6-Phosphate Dehydrogenase
|
Source: Leuconostoc mesenteroides
I.U.B.: 1.1.1.49
The Leuconostoc GPDH exhibits dual coenzyme specificity, namely NAD and NADP (Olive and Levy, Biochem., 6, 730 730, 1967). When assayed under conditions that are optimal for the particular coenzyme, the ratio of observed catalytic activity is NAD/NADP = 1.8.
Stability/Storage: The Leuconostoc mesenteroides glucose-6-phosphate dehydrogenase is a relatively stable enzyme in solution. The lyophilized and ammonium sulfate preparations are stable for 12 months when stored at 2-8°C.
Unit Definition: One Unit reduces one micromole of NAD per minute at 37°C, pH 7.8, using glucose-6-phosphate as substrate.
Glucose-6-Phosphate Dehydrogenase, Suspension
Code: ZF
Chromatographically purified. A suspension in 2.4M ammonium sulfate. Phosphohexose isomerase, phosphogluconate dehydrogenase, adenylate kinase and creatine phosphokinase contaminant activities ≤0.02%, 0.003%, 0.002% and 0.002% respectively. Store at 2-8°C.
Source: Leuconostoc mesenteroides
Minimum Activity: ≥200 NADP units per mg protein
| Cat# |
Pack Size |
Price |
|
| LS003983 |
500 un |
126.00$ |
|
| LS003985 |
5 ku |
355.00$ |
|
|
Glucose-6-Phosphate Dehydrogenase, Suspension
Code: ZFD
Chromatographically purified. Same as Code: ZF except assayed using NAD. Phosphohexose isomerase, phosphogluconate dehydrogenase, adenylate kinase and creatine phosphokinase contaminant activities ≤0.011%, 0.002%, 0.0011% and 0.0011% respectively. A suspension in 2.4M ammonium sulfate. Store at 2-8°C.
Source: Leuconostoc mesenteroides
Minimum Activity: ≥360 NAD units per mg protein
| Cat# |
Pack Size |
Price |
| LS003992 |
900 un |
126.00$ |
| LS003993 |
9 ku |
355.00$ |
|
Glucose-6-Phosphate Dehydrogenase, Lyophilized
Code: ZFL
Chromatographically purified. Phosphohexose isomerase, phosphogluconate dehydrogenase, adenylate kinase and creatine phosphokinase contaminant activities ≤0.02%, 0.003%, 0.002% and 0.002% respectively. A lyophilized powder. Store at 2-8°C.
Source: Leuconostoc mesenteroides
Minimum Activity: ≥200 NADP units per mg protein
| Cat# |
Pack Size |
Price |
| LS003981 |
1 ku |
152.00$ |
| LS003980 |
10 ku |
590.00$ |
|
Glucose-6-Phosphate Dehydrogenase, Lyophilized
Code: ZFLD
Chromatographically purified. Same as Code: ZFL except assayed using NAD. Phosphohexose isomerase, phosphogluconate dehydrogenase, adenylate kinase and creatine phosphokinase contaminant activities ≤0.011%, 0.002%, 0.0011% and 0.0011% respectively. A lyophilized powder. Store at 2-8°C.
Source: Leuconostoc mesenteroides
Minimum Activity: ≥360 NAD units per mg protein
| Cat# |
Pack Size |
Price |
| LS003997 |
2 ku |
152.00$ |
| LS003998 |
18 ku |
590.00$ |
|
| Catalog # |
Description
|
Price
|
| NaR-PkY |
Superior Stock YNaR1
freeze-dried, One vial, 1 unit/vial
+ NADH for 20-50 assays,
freeze-dried, One vial
|
75.00$ |
| NaR-PkAt |
Superior Stock AtNaR2
freeze-dried, One vial, 1 unit/vial
+ NADH for 20-50 assays,
freeze-dried, One vial
|
75.00$ |
|




